Difference between revisions of "Volatiles"
Jarogers2001 (talk | contribs) (sandworm image resized at 600px to fix overlap with TOC) |
m (Requesting Scientifiction.org fork of this article; formatting) |
||
| Line 1: | Line 1: | ||
[[Image:Sandworm01.jpg|thumb|600px|Sandworm proposal by [[User:Jriley|Tom Riley]], Side View]] | [[Image:Sandworm01.jpg|thumb|600px|Sandworm proposal by [[User:Jriley|Tom Riley]], Side View]] | ||
| − | + | <BR clear="all"/> | |
| + | {{Fork2sf}} | ||
==Volatile Recovery on the Moon== | ==Volatile Recovery on the Moon== | ||
Revision as of 06:03, 4 June 2007
It is requested that a fork of this article be installed to Scientifiction.org.
Contents
Volatile Recovery on the Moon
Long term lunar settlement, not to mention going on to Mars, will depend on successful extraction of volatile substances from the Moon.
Volatiles, The Key to Settlement
The primary resource of value to humans on the Moon is the volatile components of found in the regolith. These are all the components that are gases at room temperature. The volatiles have been deposited in the top layers of the Moon's surface by the solar wind over geologic time. It contains material useful for rocket fuel, for making air to breath, and for industrial operations.
The various constituents of the volatiles vary from place to place on the Moon. A mining area with high abundances will be a major access for any settlement site. The major components are:
- Hydrogen
- Helium4 with a trace amount of Helium3
- Nitrogen
- Inert gases, Neon and Argon]]
- Carbon compounds: Carbon Dioxide, Carbon Monoxide, and Methane
Additional processing can also extract water and oxygen.
Apollo Regolith Testing
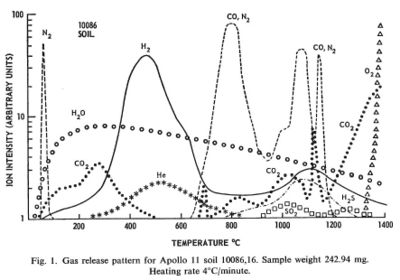
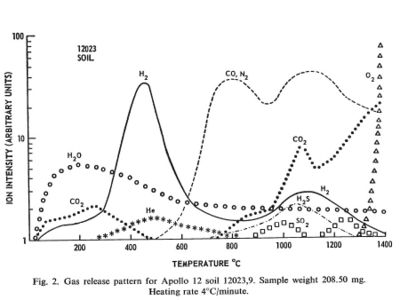
Regolith samples from Apollo 11 and Apollo 12 were returned to Earth and analyzed for the volatiles they emitted when heated in a vacuum.
The regolith samples were exposed to Earth air and may have picked up some volatiles in transport. Lunar regolith is regularly subject to heating from the sun to at least 150 C. Any volatiles coming out below that temperature was taken to be from Earth.
Between the temperatures of 150 C and 700 C substantial amounts of hydrogen, water, carbon dioxide, and helium came off the samples. In this temperature range the molecules must have been adsorbed on the surface of crystal grains and not bound in chemical compounds. These temperatures are probably achievable by the use of concentrated solar energy.
Between the temperatures of 700 C and 1400 C additional volatile materials come off including substantial amount so nitrogen, carbon monoxide, hydrogen sulfide, and finally oxygen. These volatiles probably represent the breakdown of more complex compounds. These temperatures are routinely achieved in industrial processes on Earth. The volatile output can probably be increased by providing a reducing atmosphere, such as hot hydrogen, and by the presence of a catalyst, such as platinum.
[[1]] Gibson, E. K., Jr.; Johnson, S. M., Thermal Analysis-Inorganic Gas Release of Lunar Samples
Processing Regolith
A major enterprise of any lunar settlement will be the processing of lunar regolith for Volatile Scavenging. In our stories this is done with very large vehicles that crawl across the landscape milling out a trench three meters deep and up to 23 meters wide. The Sandworms are described in Sandworms. Some of these trenches are then used as sites for buildings.
The primary uses of the volatiles are:
- Lunar use for breathable atmosphere
- Spaceship atmosphere
- Rocket fuel and oxidizer
- Industrial stocks
It is necessary to process only a few tens of kilograms of regolith a shift to replace atmosphere loses in an early small station. A much higher rate of processing will be needed to produce air to grow the settlement and to produce fuel and oxidizer for return flights to the Earth or for trips on to Mars.
Helium 3, Special Considerations
Helium 3 is a special case. As detailed in Harrison Schmitt's book "Return to the Moon", it is the only lunar resource worth shipping back to Earth. In the long term, it could be used in fusion power plants on Earth as a source of clean, non-carbon energy. This type of power plant is currently under development, but is not yet near commercial operation.
Helium 3 is very rare on Earth and only a minor component of the lunar regolith volatiles. To refine enough to feed a power plant for a medium size city would require processing regolith at the rate of about 50 tons an hour. Hence the large size of the Sandworms in our stories. A commercial operation of this size on Earth would not be considered a very large facility, but we are talking about the Moon.
Cooking out the O2
The easiest source of oxygen on the Moon is a Titanium-Iron-Oxygen mineral called ilmenite (FeTiO3). It is a straight forward process to magnetically beneficiate this mineral while handling the regolith fines in the Sandworms.
The magnetically beneficiated material is valuable as an ore concentrate and can be robotically handled. It is placed in a pressure vessel with an atmosphere of hydrogen, which is the largest component of the volatile extraction. The vessel is then solar heated to about 1200 C for 20 minutes. The result is that the ore is reduced to iron, titanium dioxide, and water. The water comes off as a vapor and can be electrically split into hydrogen and oxygen. This process appears to be the least energetic approach for obtaining substantial amounts of water and oxygen on the Moon.
The solids left from this process will remain a good titanium and iron ore. This material may become a valuable asset for later settlers. In our stories it is first used to make for ceramic tiles for flooring.
Ice at the Poles
Two missions so far (Clementine and Lunar Prospector) have produced evidence for hydrogen in the polar regions of the Moon. The present theory is that it is in the form of water ice particles mixed with the regolith in permanently shadowed craters. The amount and properties of this resource are not currently known, but we soon will have definitive data from the Lunar Reconnaissance Orbiter mission (LRO) by 2010.
Even if substantial amounts of water are present in the polar traps, it may be very difficult to mine. The very nature of the traps is that they have no access to solar power at all and they are at the bottoms of deep craters. For a settlement to take advantage of this resource it will need to be on near by high ground and work the area robotically.
This will be very difficult work at the cryogenic temperatures in the traps. It is not clear whither simply working the sunlit uplands, with plenty of solar power but only a trace of volatiles, is not the better idea. We will know soon.